This is from Canvas
This is a trial – Heading 2
paragraph
Heading 3
some text
Heading 4
some text. some text that’s bolded. Some text that is underlined.
Numbered list:
- number 1
- number 2
Bullet list:
- Bullet 1
- Bullet 2
| Column 1 | Column 2 centered |
|---|---|
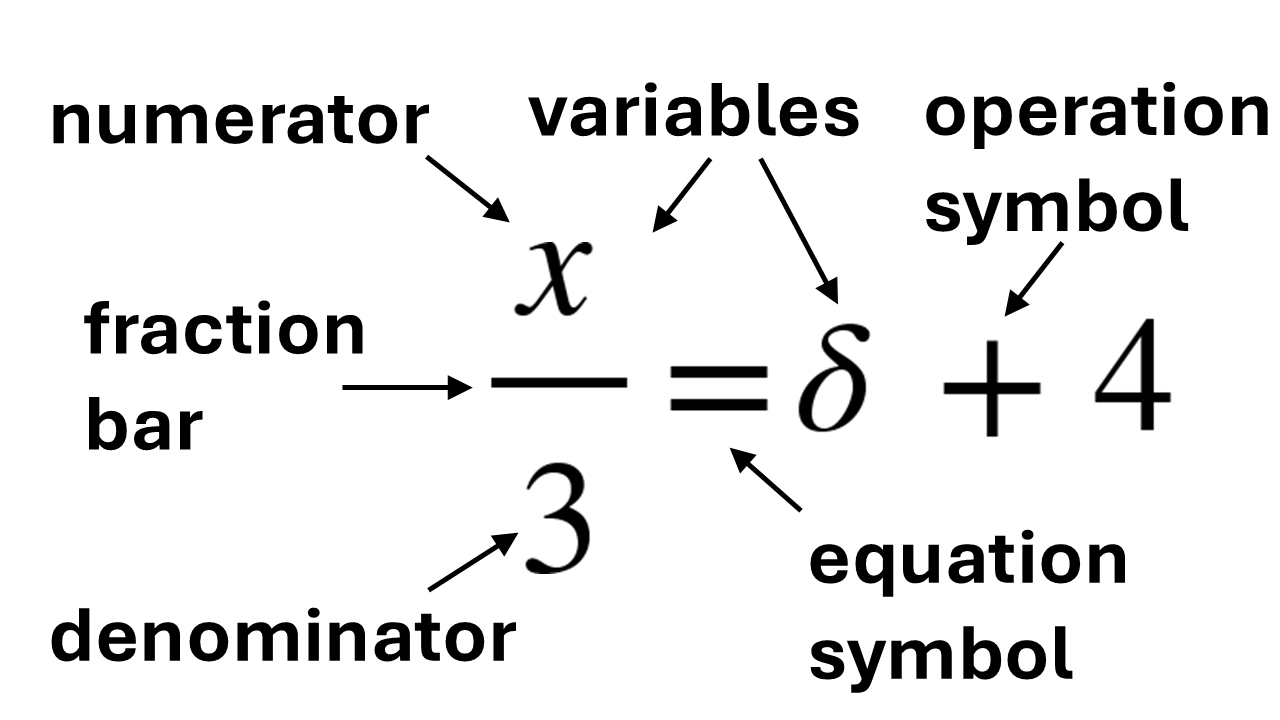
Exponential Notation
This is exponential notation where the small number written above and to the right of the base (superscript), indicating how many times the base is multiplied by itself. (example: 23 = 2 x 2 x 2)
| Exponential Number | What is happening | Ordinary Number |
|---|---|---|
| 1 x 106 | 10 x 10 x 10 x 10 x 10 x 10 | 1,000,000 |
| 1 x 103 | 10 x 10 x 10 | 1,000 |
| 1 x 102 | 10 x 10 | 100 |
| 1 x 101 | 10 | 10 |
| 1 x 100 | 1 (anything raised to 0 power is 1) | 1 |
| 1 x 10-1 | 0.1 | |
| 1 x 10-2 | 0.01 | |
| 1 x 10-3 | 0.001 | |
| 1 x 10-6 | 0.000 001 |
Scientific Notation
This is standard scientific notation where only significant digits are shown and the decimal is after the first digit. This is followed by a multiplication sign, and then 10, and then the power of 10 in superscript. (example: 3.250 x 103 = 3.250 x 10 x 10 x 10 = 3,250)
Metric Units
|
What you are measuring |
Base Unit |
Symbol |
|---|---|---|
|
Length |
meter |
m |
|
Mass |
kilogram |
kg |
|
Volume |
liter |
L |
|
Temperature |
kelvin |
K |
|
Time |
seconds |
s |
|
Amount of something |
mole |
mol |
Metric Prefixes
| Prefix | Abbreviation | Meaning | Example |
|---|---|---|---|
| tera | T | 1,000,000,000,000 or 1012 | 1 teragram (Tg) = 1012 g |
| giga | G | 1,000,000,000 or 109 | 1 gigameter (Gm) = 109 m |
| mega | M | 1,000,000 or 106 | 1 megaliter (ML) = 106 L |
| kilo | k | 1,000 or 103 | 1 kilosecond (ks) = 1,000 s |
| hecto | h | 100 or 102 | 1 hectomole (hmol) = 100 mol |
| deka | da | 10 or 101 | 1 dekakelvin (daK) = 10 kelvin |
| 1 | 1 gram (g) or any unit | ||
| deci | d | 1/10 or 0.1 or 10-1 | 1 decimeter (dm) = 0.1 m |
| centi | c | 1/100 or 0.01 or 10-2 | 1 centiliter (cL) = 0.01 L |
| milli | m | 1/1,000 or 0.001 or 10-3 | 1 millisecond (ms) = 0.001 s |
| micro | m | 1/1,000,000 or 10-6 | 1 micromole (mmol) = 10-6 mol |
| nano | n | 1/1,000,000,000 or 10-9 | 1 nanogram (ng) = 10-9 g |
| pico | p | 1/1,000,000,000,000 or 10-12 | 1 picokelvin (pK) = 10-12 K |
Common Conversions
| Conversion Factors / Conversion Rates | Description (can convert the reverse as well) |
|---|---|
| 1 milliliter (mL) = 1 centimeter cubed (cm3) or (cm x cm x cm) | use to convert a milliliter (mL) volume to a centimeter cubed (cm3) volume |
| 1 liter (L) = 1000 centimeter cubed (cm3) or (cm x cm x cm) | use to convert a liter (L) volume to a centimeter cubed (cm3) volume |
| 1 inch (in) = 2.54 centimeters (cm) | use to convert from inches (standard system) to centimeters (metric system) |
| 1 yard (yd) = 0.9144 meters (m) | use to convert from yard (standard system) to meters (metric system) |
| 454 grams (g) = 1 pound (lb) | use to convert grams (metric system) to pounds (standard system) |
| 0.946 liters (L) = 1 quart (qt) | use to convert liters (metric system) to quarts (standard system) |
| 1 mole = 6.022 x 1023 things (usually atoms or molecules) | can use this to convert in and out of moles (moles to atoms or molecules) |
Test1




Test 2
Introduction
Chemistry requires the use of various math operations including algebra. A common reason why students struggle to be successful in chemistry is their math skills are not sufficient for chemistry. Students then have to learn complex chemistry concepts and calculations as well as math at the same time. This will help students learn the math fundamentals to be successful in chemistry.
Contents
· Part A: Identification of the parts of an equation
o Example A1: Identification of the parts of an equation
§ Practice A1: Identification of the parts of an equation
§ Practice A2: Infer the “hidden” parts of an equation
· Part B: Isolate a variable
o Example B1: Identify the math operation and opposite operation to isolate the variable
§ Practice B1: Identify the math operation and opposite operation to isolate the variable
o Example B2: Solve for a variable (addition/subtraction)
§ Practice B2: Solve for a variable
o Example B3: Solve for a variable (addition/subtraction)
§ Practice B3: Solve for a variable
o Example B4: Solve for a variable (multiplication/division)
§ Practice B4: Solve for a variable
o Example B5: Solve for a variable (multiplication/division)
§ Practice B5: Solve for a variable
· Multiple operations
o Example B6: Solve for a variable (exponents/roots)
§ Practice B6: Solve for a variable
o Example B7: Solve for a variable (exponents/roots)
§ Practice B7: Solve for a variable
· Multiple operations
o Example B8: Solve for a variable (variable in exponent/logarithms)
§ Practice B8: Solve for a variable
o Example B9: Solve for a variable (natural log)
§ Practice B9: Solve for a variable
o Example B10: Solve for a variable (ex)
§ Practice B10: Solve for a variable
· Multiple operations
· Exercises
· Answer Key to Practice Problems
· Answer Key to Exercises
Part A: Identification of the parts of an equation
In chemistry we often use variables. A variable is usually a letter or symbol used in equations that can have different values. Figure 1A shows an equation with name labels in the left figure below. If there is no fraction bar, the number is considered to be a numerator with an imaginary fraction bar and a 1 in the denominator (Figure 1B).
Example A1: Identification of the parts of an equation
|
A |
B |
Figure 1: (A) Figure showing various names of objects in an equation (left). (B) The same equation but including the imaginary fraction bars and the numbers 1 in the denominators (right).
Practice A1: Identify the parts of an equation
|
Label the following parts of this equation: · Numerators · Denominators · Equal sign · Variables · Fraction Bar · Operation symbols |
|
Practice A2: Infer the “hidden” parts of an equation
|
Write-in the imaginary fraction bars and imaginary 1’s in the denominators
|
|
Part B: Isolate a variable
We routinely solve for variables in chemistry. This means get the variable by itself on one side of the equation symbol. This also means get the variable on top of the fraction bar in the numerator position. How do we get the variable “y” by itself on one side of the equation in the numerator position? To do this you must do the opposite of the operation it currently is associated with. Also, for the equation to remain correct, you must perform whatever opposite operation is needed to both sides of the equal sign. Table 1 shows a column of math operations on the left and the corresponding opposite operation.
|
Math Operation |
Opposite of Operation |
|
Addition |
Subtraction |
|
Subtraction |
Addition |
|
Multiplication (in the numerator) |
Division |
|
Division (in the denominator) |
Multiplication |
|
x2 (Squared) |
Square root or raise to the 1/2 power |
|
X3 (Cubed) |
Cubed root or raise to the 1/3 power |
|
anythingx (exponent) |
log (x) (logarithm) |
|
ln (x) (natural log) |
ex (Euler’s number) |
Table 1: A list of math operations (left) as well as the corresponding opposite math operation (right).
Example B1: Identify the math operation and opposite operation to isolate the variable
|
Equation |
Operation that the variable is involved in |
Opposite operation to isolate the variable |
|
<img src="https://latex.codecogs.com/svg.image?&space;y-3=6" title=" y-3=6" /> |
3 is being subtracted from y |
You must add 3 |
|
|
9 is being added to n |
You must subtract 9 |
|
|
7 is being multiplied by x |
You must divide by 7 |
|
|
|
|
|
|
X is being raised to the 3rd power |
You must take the cubed root of X or raise to the 1/3 power |
|
|
X is being squared |
You must raise this to |
|
|
|
|
|
ln (x) |
|
|
Practice B1: Identify the math operation and opposite operation to isolate the variable
|
Equation |
Operation that the variable is participating in |
Opposite operation to isolate the variable |
|
|
3 is being subtracted from y |
You must add 3 |
|
|
9 is being added to n |
You must subtract 9 |
|
|
7 is being multiplied by x |
You must divide by 7 |
|
|
|
|
|
|
|
|
|
|
|
|
Example B2: Solve for a variable (addition/subtraction)
|
Problem: |
|
Solve for “y” |
|
|
|
|
|
Equation: |
|
|
|
Step 1: |
+ |
Add 3 to both sides of the equation. |
|
Explanation: |
|
Since 3 is initially being subtracted from “y” on the left size, 3 must be added to “y” (addition is the opposite operation of subtraction) to remove the “ – 3 “ on the left side and get the variable “y” by itself. Add 3 to both sides of the equation because the same operation must be performed to both sides of the equation for the equation to remain true. |
|
|
|
On the left side of the equation symbol, +3 and -3 equal 0 |
|
|
|
Since subtracting or adding 0 to something doesn’t change it’s value, you can just leave 0 out. |
|
Problem: |
|
Solve for “y” |
|
|
|
|
|
Equation: |
|
|
|
Step 1: |
|
Add 3 to both sides of the equation. |
|
Explanation: |
|
Since 3 is initially being subtracted from “y” on the left size, 3 must be added to “y” (addition is the opposite operation of subtraction) to remove the “ – 3 “ on the left side and get the variable “y” by itself. Add 3 to both sides of the equation because the same operation must be performed to both sides of the equation for the equation to remain true. |
|
|
|
On the left side of the equation symbol, +3 and -3 equal 0 |
|
|
|
Since subtracting or adding 0 to something doesn’t change it’s value, you can just leave 0 out. |
Practice B2: Solve for a variable
Example B3:
|
Problem: |
|
Solve for “n” |
|
|
|
|
|
Equation: |
|
|
|
Step 1: |
|
Subtract 9 from both sides of the equation. |
|
Explanation: |
|
Since 9 and “n” are being added together, 9 must be subtracted from the right side (subtraction is the opposite operation of addition) to remove the 9 on the right side and get the variable “n” by itself. Subtract 9 from both sides of the equation because the same operation must be performed to both sides of the equation for the equation to remain true. |
|
|
|
On the right side of the equation symbol, +9 and -9 equal 0 |
|
|
|
Since subtracting or adding 0 to something doesn’t change it’s value, you can just leave 0 out. |
Example B4:
Part B: Get a variable into the numerator position
Text
Part C: Multiple operations to solve for a variable
Text
Lesson 3: Simplifying
Text
Notes:
· Practice in each section
o Y=mx+b
§ Curved lines with e
o PV=nRT
o Temp conversions
· Do all operations
· Do multi-variable operations
o Have to distribute for various things
o Pemdas???
Exercises:
Lesson 1:
Lesson 2:
Lesson 3:
Introduction
Chemistry requires the use of various math operations including algebra. A common reason why students struggle to be successful in chemistry is their math skills are not sufficient for chemistry. Students then have to learn complex chemistry concepts and calculations as well as math at the same time. This will help students learn the math fundamentals to be successful in chemistry.
Contents
- Part A: Identification of the parts of an equation
- Example A1: Identification of the parts of an equation
- Practice A1: Identification of the parts of an equation
- Practice A2: Infer the “hidden” parts of an equation
- Example A1: Identification of the parts of an equation
- Part B: Isolate a variable
- Example B1: Identify the math operation and opposite operation to isolate the variable
- Practice B1: Identify the math operation and opposite operation to isolate the variable
- Example B2: Solve for a variable (addition/subtraction)
- Practice B2: Solve for a variable
- Example B3: Solve for a variable (addition/subtraction)
- Practice B3: Solve for a variable
- Example B4: Solve for a variable (multiplication/division)
- Practice B4: Solve for a variable
- Example B5: Solve for a variable (multiplication/division)
- Practice B5: Solve for a variable
- Example B1: Identify the math operation and opposite operation to isolate the variable
- Multiple operations
- Example B6: Solve for a variable (exponents/roots)
- Practice B6: Solve for a variable
- Example B7: Solve for a variable (exponents/roots)
- Practice B7: Solve for a variable
- Multiple operations
- Example B8: Solve for a variable (variable in exponent/logarithms)
- Practice B8: Solve for a variable
- Example B9: Solve for a variable (natural log)
- Practice B9: Solve for a variable
- Example B10: Solve for a variable (ex)
- Practice B10: Solve for a variable
- Example B8: Solve for a variable (variable in exponent/logarithms)
- Multiple operations
- Exercises
- Answer Key to Practice Problems
- Answer Key to Exercises
Part A: Identification of the parts of an equation
In chemistry we often use variables. A variable is usually a letter or symbol used in equations that can have different values. Figure 1A shows an equation with name labels in the left figure below. If there is no fraction bar, the number is considered to be a numerator with an imaginary fraction bar and a 1 in the denominator (Figure 1B).
Example A1: Identification of the parts of an equation
| A | B |
Figure 1: (A) Figure showing various names of objects in an equation (left). (B) The same equation but including the imaginary fraction bars and the numbers 1 in the denominators (right).
Practice A1: Identify the parts of an equation
| Label the following parts of this equation: NumeratorsDenominatorsEqual signVariablesFraction BarOperation symbols | |
Practice A2: Infer the “hidden” parts of an equation
| Write-in the imaginary fraction bars and imaginary 1’s in the denominators | |
Part B: Isolate a variable
We routinely solve for variables in chemistry. This means get the variable by itself on one side of the equation symbol. This also means get the variable on top of the fraction bar in the numerator position. How do we get the variable “y” by itself on one side of the equation in the numerator position? To do this you must do the opposite of the operation it currently is associated with. Also, for the equation to remain correct, you must perform whatever opposite operation is needed to both sides of the equal sign. Table 1 shows a column of math operations on the left and the corresponding opposite operation.
| Math Operation | Opposite of Operation |
| Addition | Subtraction |
| Subtraction | Addition |
| Multiplication (in the numerator) | Division |
| Division (in the denominator) | Multiplication |
| x2 (Squared) | Square root or raise to the 1/2 power |
| X3 (Cubed) | Cubed root or raise to the 1/3 power |
| anythingx (exponent) | log (x) (logarithm) |
| ln (x) (natural log) | ex (Euler’s number) |



