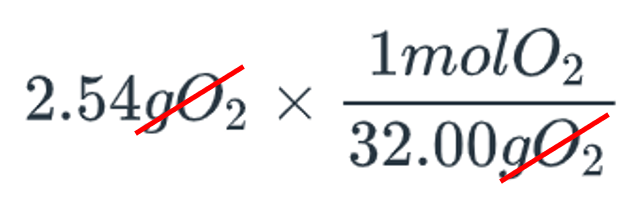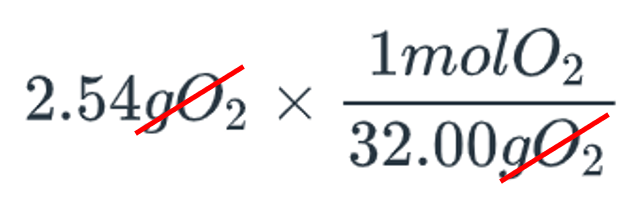
Exponential Notation
block
inline
Pasted into Canvas from screenshot in PowerPoint and then alt text added into Canvas
Pasted into comstgermain and then image address copied and put into Canvas and then alt text added into Canvas
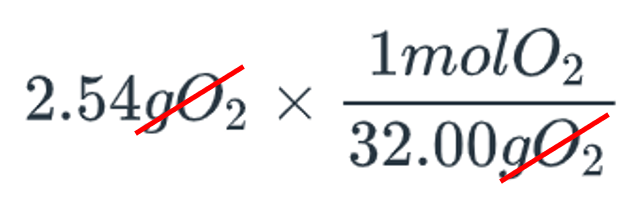
| Verbal Explanation | Equations and Work |
| the value you want to convert, 2.54 grams of O2 multiplied by 1 mol of O2 divided by 32.00 grams of O2 | |
| the grams of O2 cancel out on the top and bottom of the equations | 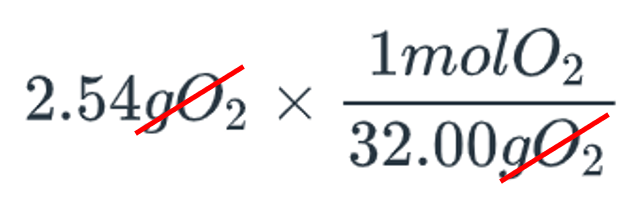 |
This is exponential notation where the small number written above and to the right of the base (superscript), indicating how many times the base is multiplied by itself. (example: 23 = 2 x 2 x 2)
| Exponential Number | What is happening | Ordinary Number |
|---|---|---|
| 1 x 106 | 10 x 10 x 10 x 10 x 10 x 10 | 1,000,000 |
| 1 x 103 | 10 x 10 x 10 | 1,000 |
| 1 x 102 | 10 x 10 | 100 |
| 1 x 101 | 10 | 10 |
| 1 x 100 | 100 | 1 |
| 1 x 10-1 | 0.1 | |
| 1 x 10-2 | 0.01 | |
| 1 x 10-3 | 0.001 | |
| 1 x 10-6 | 0.000 001 |
Scientific Notation
This is standard scientific notation where only significant digits are shown and the decimal is after the first digit. This is followed by a multiplication sign, and then 10, and then the power of 10 in superscript. (example: 3.250 x 103 = 3.250 x 10 x 10 x 10 = 3,250)
Metric Units
|
What you are measuring |
Unit (with no prefix) |
Symbol |
|---|---|---|
|
Length |
meter |
m |
|
Mass |
gram |
g |
|
Volume |
liter |
L |
|
Temperature |
kelvin |
K |
|
Time |
seconds |
s |
|
Amount of something (6.022 x 1023 things) |
mole |
mol |
Metric Prefixes
Metric prefixes are just words that mean a certain number. These can be used with ANY UNIT.
| Prefix | Abbreviation | Meaning | Examples | More Examples |
|---|---|---|---|---|
| tera | T | 1,000,000,000,000 or 1012 | 1 Tm = 1012 m 1 Tg = 1012 g 1 TL = 1012 L |
1 TK = 1012 K 1 Ts = 1012 s 1 Tmol = 1012 mol |
| giga | G | 1,000,000,000 or 109 | 1 pm = 10-12 m 1 pg = 10-12 g 1 pL = 10-12 L |
1 pK = 10-12 K 1 ps = 10-12 s 1 pmol = 10-12 mol |
| mega | M | 1,000,000 or 106 | 1 pm = 10-12 m 1 pg = 10-12 g 1 pL = 10-12 L |
1 pK = 10-12 K 1 ps = 10-12 s 1 pmol = 10-12 mol |
| kilo | k | 1,000 or 103 | 1 pm = 10-12 m 1 pg = 10-12 g 1 pL = 10-12 L |
1 pK = 10-12 K 1 ps = 10-12 s 1 pmol = 10-12 mol |
| hecto | h | 100 or 102 | 1 pm = 10-12 m 1 pg = 10-12 g 1 pL = 10-12 L |
1 pK = 10-12 K 1 ps = 10-12 s 1 pmol = 10-12 mol |
| deka | da | 10 or 101 | 1 pm = 10-12 m 1 pg = 10-12 g 1 pL = 10-12 L |
1 pK = 10-12 K 1 ps = 10-12 s 1 pmol = 10-12 mol |
| 1 |
1 pm = 10-12 m |
1 pK = 10-12 K |
||
| deci | d | 1/10 or 0.1 or 10-1 | 1 pm = 10-12 m 1 pg = 10-12 g 1 pL = 10-12 L |
1 pK = 10-12 K 1 ps = 10-12 s 1 pmol = 10-12 mol |
| centi | c | 1/100 or 0.01 or 10-2 | 1 pm = 10-12 m 1 pg = 10-12 g 1 pL = 10-12 L |
1 pK = 10-12 K 1 ps = 10-12 s 1 pmol = 10-12 mol |
| milli | m | 1/1,000 or 0.001 or 10-3 |
1 mm = 10-3 m
|
1 mK = 10-3 K 1 ms = 10-3 s 1 mmol = 10-3 mol |
| micro | 1/1,000,000 or 10-6 |
1 |
1 1 1 |
|
| nano | n | 1/1,000,000,000 or 10-9 | 1 nm = 10-9 m 1 ng = 10-9 g 1 nL = 10-9 L |
1 nK = 10-9 K 1 ns = 10-9 s 1 nmol = 10-9 mol |
| pico | p | 1/1,000,000,000,000 or 10-12 | 1 pm = 10-12 m 1 pg = 10-12 g 1 pL = 10-12 L |
1 pK = 10-12 K 1 ps = 10-12 s 1 pmol = 10-12 mol |
Common Conversions
| Conversion Factors / Conversion Rates | Description (can convert the reverse as well) |
|---|---|
| 1 milliliter (mL) = 1 centimeter cubed (cm3) or (cm x cm x cm) | use to convert a milliliter (mL) volume to a centimeter cubed (cm3) volume |
| 1 liter (L) = 1000 centimeter cubed (cm3) or (cm x cm x cm) | use to convert a liter (L) volume to a centimeter cubed (cm3) volume |
| 1 inch (in) = 2.54 centimeters (cm) | use to convert from inches (standard system) to centimeters (metric system) |
| 1 yard (yd) = 0.9144 meters (m) | use to convert from yard (standard system) to meters (metric system) |
| 454 grams (g) = 1 pound (lb) | use to convert grams (metric system) to pounds (standard system) |
| 0.946 liters (L) = 1 quart (qt) | use to convert liters (metric system) to quarts (standard system) |
| 1 mole = 6.022 x 1023 things (usually atoms or molecules) | can use this to convert in and out of moles (moles to atoms or molecules) |
How to Convert Anything in World in 3 Steps:
Step 1: Find the conversion rates.
Almost any two values with units with an equals sign between them can be a conversion rate or conversion factor (same thing, different name). You can find conversion rates in this tutorial, in your book, on the internet, on food menus, etc. Directly below is a generic version of a conversion rate
Below are some conversion rates.
| Number | Conversion Rate | Description |
| 1 | USD$1.00 = MXN$18.67 | This means 1 US dollar is equivalent to 18.67 Mexican pesos. This will allow you to convert from pesos to dollars and dollars to pesos. |
| 2 | $0.99 = 2 tacos | This means 2 tacos is equivalent to 0.99 of a dollar (99 cents). This will allow you to convert from tacos to dollars and dollars to tacots. |
| 3 | 1000 g = 1 kg | This means 1000 grams is equal to 1 kilogram. This will allow you to convert from grams to kilograms and kilograms to grams. |
| 4 | 1 nmol = 10-9 mol | This means 1 nanomole is equal to 0.000 000 001 moles. This will allow you to convert from moles to nanomoles and nanomoles to moles. |
| 5 | This means 12.01 grams of carbon is equal to 1 mole of carbon. This will allow you to convert from grams of carbon to moles of carbon and from moles of carbon to grams of carbon. Conversion rates can be written as a fraction. More information is below. |
Step 2: Write the conversion rate as a fractions
Any of the conversion rates above can be written in different forms which include fractions.
| Number | Form: one equals the other | Form: one fraction | Form: the other fraction |
| 1 | USD$1.00 = MXN$18.67 | ||
| 2 | $0.99 = 2 tacos | ||
| 3 | 1000 gram (g) = 1 kilogram (kg) | ||
| 4 | 1 nanomole (nmol) = 10-9 mole (mol) | ||
| 5 | 12.01 g C = 1 mol C |
Step 3: Multiply what you are trying to convert by the fraction.
Use the conversion rate above to multiply by the value you are trying to convert. Use the fraction that has the same unit as the original value on the bottom. Cancel out anything that is the same on top of the fraction line and the bottom of the fraction line.
| Number | Original Value (what you are trying to convert) | Conversion Rate | Problem | Work (see the units cancelling out) | Answer (any units that don’t cancel carry over to the answer) |
| 1 | USD$ 5.00 | MXN$ 93.35 | |||
| 2 | 4 tacos | $1.98 | |||
| 3 | 327 grams | 0.327 kg | |||
| 4 | 4.1 x 10-6 nmol | 4.1 x 10-15 mol | |||
| 5 | 47.3 g C | 3.94 mol C |